Successful Lysis of Thrombus with Tissue Plasminogen Activator in a Bidirectional Glenn on Venoarterial Extracorporeal Life Support
Lindsay Ryerson, Cameron Seaman, Paula Holinski, Mary Bauman and Patti Massicotte
1Pediatric Cardiac Intensive Care Unit, Stollery Children’s Hospital, Edmonton, Canada
2Division of Pediatric Cardiology, Stollery Children’s Hospital, Edmonton, Canada
3Department of Pediatrics, Stollery Children’s Hospital, Edmonton, Canada
- *Corresponding Author:
- Lindsay Ryerson
Pediatric Cardiac Intensive Care Unit, Stollery Children’s Hospital
3A3.19 WMC, 8440 112 St, Edmonton
AB, T6G 2B7, Canada
Tel: +17804073396
E-mail: ryerson@ualberta.ca
Received date: October 28, 2016; Accepted date: November 22, 2016; Published date: November 29, 2016
Citation: Lindsay Ryerson MD, Seaman C,Paula Holinski MD, et al. Successful Lysis of Thrombus with Tissue Plasminogen Activator in a Bidirectional Glenn on Venoarterial Extracorporeal Life Support. Insights Pediatr Cardiol 2016, 1:1.
Copyright: © 2016 Lindsay Ryerson MD. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
This case study reports a seven month male who was cannulated for venoarterial extracorporeal life support for hypoxemia due to thrombosis of the bidirectional Glenn circulation. He received low dose tissue plasminogen activator (tPa) (0.03 mg/kg/hr) on ECLS with complete thrombolysis. tPa infusion was complicated by mediastinal bleeding.
Keywords
Pediatric; ECMO; Thrombosis; Anticoagulation; Congenital heart disease; Thrombolysis
Case Report
This case report describes a 7 kg male with double inlet right ventricle, transposition of the great arteries and pulmonary atresia admitted to Stollery Children’s Hospital, Edmonton, Alberta for bidirectional Glenn (BDG) and patch augmentation of the left pulmonary artery (LPA). The patient had undergone three previous operations, initially a right modified Blalock-Taussig shunt (3.5mm), followed by several attempts to recruit the LPA by bovine pericardial patch. He was palliated with a BDG and LPA autologous pericardial patch. The LPA thrombosed accepts for its intraparenchymal portion. Given the presence of ongoing hypoplasia and acute thrombosis of the LPA, the decision was made to place a central shunt with the aim of securing pulmonary blood flow to the left lung and augmenting growth of peripheral vasculature. A 4.0 mm Gore-Tex central shunt from the ascending aorta to LPA was placed which subsequently thrombosed. The child proceeded to develop both chylothorax and mediastinitis. A peripherally inserted central cannula (PICC) was placed in the right arm with the tip in the SVC for total parenteral nutrition and antibiotics. Forty-eight hours post mediastinal washout, the patient became acutely hypoxemic and had a cardiopulmonary arrest. Although return of spontaneous circulation was restablished, the patient was cannulated centrally (right atrium and ascending aorta) for VA ECLS for hyperlactatemia. The ECLS run was complicated by circuit thrombosis despite therapeutic anti-Xa levels. Confirmatory testing for heparin-induced thrombocytopenia by lumi-aggregometry was negative. He was decanulated after 43 hours, however two days later the patient thrombosed his BDG circulation and required repeat central ECLS cannulation for hypoxemia.
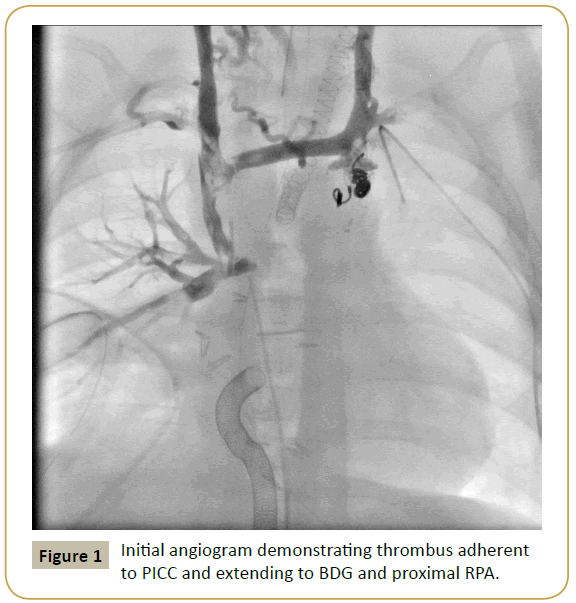
Given persistent development of circuit thrombosis while on an unfractionated heparin (UFH) infusion during the initial ECLS run, the decision was made to anticoagulate using bivalirudin. Cardiac catheterization demonstrated a large, partially occlusive thrombus in the SVC and proximal right pulmonary artery adherent to the PICC line (Figure 1). This was partially aspirated resulting in significant reduction in thrombus load. Additionally, an angiogram performed via the arterial limb of the ECLS circuit demonstrated two infra-renal thrombi in the descending aorta. The use of tissue plasminogen activator (tPa) thrombolysis was discussed and agreed upon, therefore a sheath was left in the right internal jugular vein (RIJV) for repeat angiography following tPa thrombolysis.
The patient returned to the pediatric cardiac intensive care unit (PCICU) on ECLS for administration of systemic thrombolysis. Prior to tPa initiation, fibrinogen was 1.3 g/L and platelets 49,000 × 109/L. One unit of cryoprecipitate, 100 mls of platelets, 10 U/ ml/kg of plasma was administered and bivalirudin was changed to an UFH infusion at 20 U/kg/hr because of clinical familiarity and reversibility. Following plasma bolus, a continuous infusion of plasma of 10 ml/hour was administered for the duration of the tPa infusion. tPa was infused at 0.01 mg/kg/hr (without bolus) through three different sites: RIJV sheath, right arm PICC and arterial limb of the ECLS circuit for a total dose of 0.03 mg/kg/ hr. After one hour of tPa, UFH infusion was empirically increased to 28 U/kg/hr. Target anti-Xa level was 0.3-0.5 U/ml. Empiric fibrinogen target was ≥ 2 g/L and platelets ≥ 100,000 x 109/L. Due to increased fibrin deposition in the circuit, UFH was incrementally increased to 34 U/kg/hr maintaining the target anti-Xa level. The emergency plan for bleeding was 3U of cryoprecipitate.
The patient developed mediastinal bleeding after 16 hours of systemic tPa. tPa and UFH infusions were discontinued and 3U of cryoprecipitate, 20 ml/kg of platelets and a unit of red blood cells administered. Mediastinal exploration demonstrated bleeding from the aortic cannula that was controlled with suture. Angiography on two subsequent days demonstrated significant reduction in residual thrombus in the BDG. The PICC line was pulled back to the right subclavian vein. Final angiogram showed a widely patent SVC with non-occlusive thrombus in the right upper branch pulmonary artery. There was residual thrombus in the infra-renal descending aorta. He resumed bivalirudin anticoagulation after tPa was discontinued.
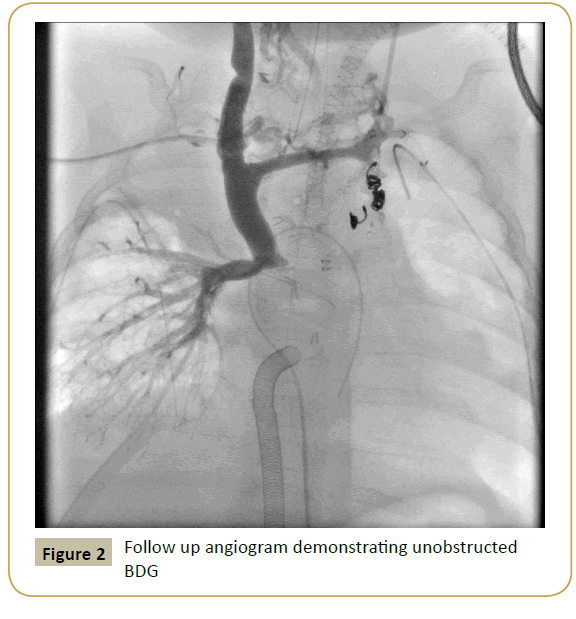
Hypoxemia prevented weaning from ECLS and as a result, a second tPa infusion at 0.01 mg/kg/hr was administered for 12 hours via the PICC line. Bivalirudin infusion was converted back to UFH during tPa infusion. Repeat angiogram demonstrated no evidence of thrombi in BDG or peripheral lung vasculature (Figure 2).
The patient was decannulated from ECLS after 183 hours. Cranial ultrasounds performed daily demonstrated no acute intracranial abnormality. Thrombophilic workup including Factor V Leiden, prothrombin gene 20210, Protein C, Protein S, antithrombin and homocysteine levels were normal. He was discharged after a seven-month hospital stay awaiting further surgical palliation to rehabilitate his LPA..
Discussion
Thrombosis of the BDG is a rare complication [1-3]. The inciting factor for this patient’s BDG thrombus was likely the PICC line; the thrombophilic workup was unremarkable. Given the BDG circulation and limited success with interventional thrombectomy, the use of systemic thrombolysis with tPa was considered this patient’s only option. To our knowledge, this is the first report of the use of tPa thrombolysis of a BDG on ECLS. tPa promotes fibrinolysis by activating plasminogen to its active form, plasmin, which results in lysis of fibrin at the site of thrombus. tPa also cleaves fibrinogen, which increases the risk of pathologic bleeding [4]. The clinical team must balance the risk of hemorrhage with the risk of persistent thrombus. This patient had complete thrombolysis, but complicated by major bleeding requiring mediastinal exploration and transfusion of multiple blood products since the patient also required plasma and platelet transfusion. There was no intracranial hemorrhage.
The optimal dose, duration and anticoagulation management for children who require tPa thrombolysis is unknown. There are no evidence-based guidelines for medical thrombolysis with tPa in children. There are reports outlining tPa regimens in pediatrics [5,6], however there is limited experience with systemic tPa infusions on ECLS with high mortality; all describe standard dosing regimens of 0.1-0.5 mg/kg/hr [7-9]. Similar efficacy is described between low dose tPa (0.01-0.06 mg/kg/hr) and standard dosing (0.1-0.5 mg/kg/hr) regimens in neonates and children with arterial and venous thrombi [5,6]. Hemorrhagic complications were not associated with tPa total dose or duration of infusion [5].
The decision to switch the patient from bivalirudin to UFH during tPa infusion was due to reversibility of UFH and more clinical experience with UFH. UFH during the v infusion was administered with an aim to prevent proximal thrombus extension and circuit thrombosis as tPa thrombolysis does not inhibit clot propagation or directly alter hypercoagulability [5]. Furthermore, concomitant administration of prophylactic UFH is not reported to increase risk of hemorrhage [5]. Plasma was administered concurrently to provide a fibrinogen source.
This case report is important as it describes a previously unreported option for medical thrombolysis in a medically complex infant on ECLS thereby avoiding an additional high-risk surgery for embolectomy. Low dose tPa infusion of 0.03 mg/kg/ hr was successful with complete thrombolysis in an infant while on ECLS. For safety, a fibrinogen threshold of minimally 1 g/L and platelet count ≥ 100,000 x 109/L should be considered to minimize bleeding complications.
References
- Zahr RA, Kirshbom PM, Kopf GS, Sainathan S, Steele MM et al. (2016) Half a century's experience with the superior cavopulmonary (classic Glenn) shunt. Ann Thorac Surg 101: 177-182.
- Pennington DG, Nouri S, Ho J, Secker-Walker R, Patel B et al. (1981) Glenn shunt: long-term results and current role in congenital heart operations. Ann Thorac Surg 31: 532-539.
- Kopf GS, Laks H, Stansel HC, Hellenbrand WE, Kleinman CS et al. (1990) Thirty-year follow-up of superior vena cava-pulmonary artery (Glenn) shunts. J Thorac Cardiovasc Surg 100: 662-670.
- Haire WD (1992) Pharmacology of fibrinolysis. Chest 101: 91S-97S.
- Wang M, Hays T, Balasa V, Bagatell R, Gruppo R, et al. (2003) Low-dose tissue plasminogen activator thrombolysis in children. J Pediat Hematol Oncol 25: 379-386.
- Manco-Johnson MJ, Grabowski EF, Hellgreen M, Kemahli AS, Massicotte MP et al. (2002) Recommendations for TPA thrombolysis in children. On behalf of the Scientific and Standardization Committee of the International Society of Thrombosis and Haemostasis. Thromb Haemost 88: 157-158.
- Garcia A, Gander JW, Gross ER, Reichstein A, Cowles RA et al. (2011) The use of recombinant tissue-type plaminogen activator in a newborn with an intracardiac thrombus developed during extracorporeal membrane oxygenation. J Ped Surg 46: 2021-2024.
- Gunnarsson B, Heard CMB, Martin DJ, Brecher ML, Steinhorn RH (2000) Successful lysis of an obstructive aortic and renal artery thrombus in a neonate on extracorporeal membrane oxygenation. J Perinatol 20: 555-557.
- Glover ML, Camacho Mt, Wolfsdorf J (1999) The use of alteplase in a newborn receiving extracorporeal membrane oxygenation. Ann Pharmacother 33: 416-419.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences